Award-Winning Chicago Personal Injury Lawyer - Securing Justice
for Illinois Injury Victims - Over $450 Million Recovered
Thousands of lawsuits have been filed against Stryker, a major medical device manufacturer, for early failure of its hip implants. These defective medical devices have caused injuries to many patients who also need to get another hip replacement surgery to take out the defective implant. Stryker is being sued by patients to receive compensation for their pain and suffering.
We’re actively investigating all Stryker suits and remain committed to helping clients like you receive fair compensation for the injuries caused by defective hip devices. Read on to learn how our reliable and experienced legal representation can help in your fight to get justice.
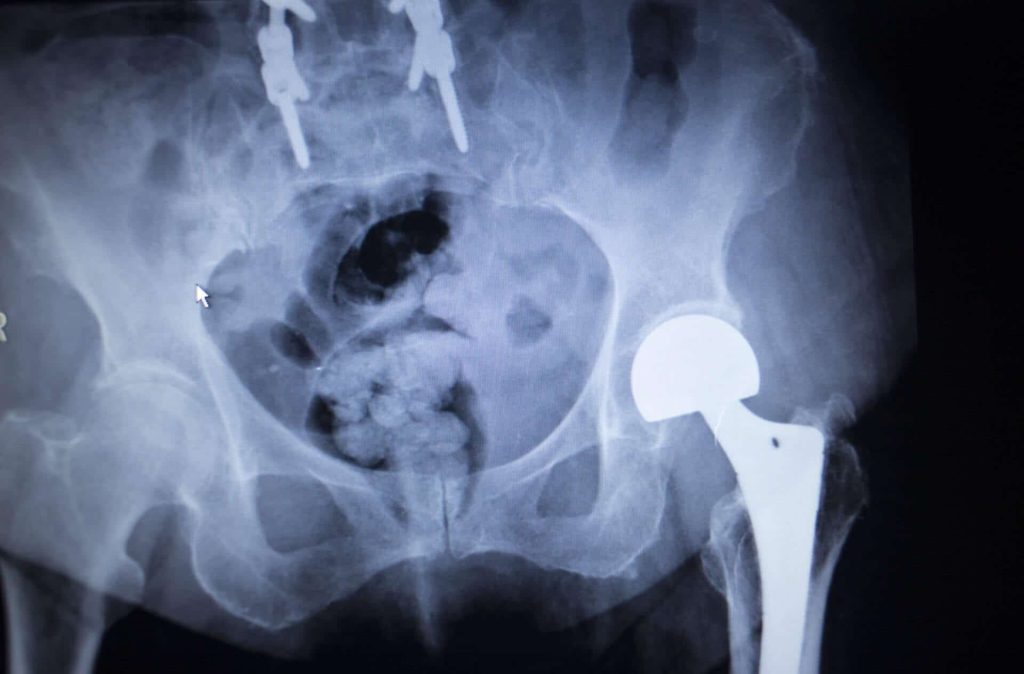
Lawsuits have been filed against Stryker Orthopaedics after it issued a voluntary recall for the Stryker ABG II hip, Stryker Rejuvenate, LFIT Anatomical CoCr V40 Femoral Head, and Tritanium Acetabular Shell devices, prompted by reports of excessive metal debris and ion generation. Stryker’s metal-on-metal implants are made from different metal alloys for enhanced strength.
However, these defective implants do not function properly, resulting in serious complications for many patients, such as pain, inflammation, loss of mobility, infection, dislocation, blood clots, and metal ions being absorbed into the bloodstream. Patients have filed lawsuits against Stryker Corporation to receive compensation for additional revision surgery, treatment, and other medical bills.
Ultimately, Stryker Corp has been the recipient of several lawsuits related to Rejuvenate and ABG II hip replacement systems because of their defective designs and associated health risks. Some people argue that Stryker failed to carry out clinical studies to confirm the safety and effectiveness of the Rejuvenate and ABG II hip systems before making these devices available for sale.
Thousands of lawsuits have been filed against Stryker by people who received one of its defective hip replacements. Settlements have been reached in many Stryker cases, but there are pending lawsuits in state and federal courts, including both individual and class action lawsuits.
A large number of cases were filed in the New Jersey state courts, as Stryker has its manufacturing facility there. Cases have also been filed in Michigan, where Stryker is based.
Some cases are still ongoing even though many plaintiffs have received settlements. Stryker has also been facing Tritanium Acetabular Shell lawsuits since 2019, with no settlements in these cases yet.
There wasn’t a class action lawsuit against Stryker; rather, the company faced multicounty and multi-district litigation (MDL) due to its defective devices. The initial $1.4 billion 2014 settlement for the Stryker ABG II and Rejuvenate hip products was reached in the New Jersey multicounty litigation, and the MDL presided over by Judge Franklin L. Noel for the District of Minnesota.
The pending lawsuits against LFIT V40 have been consolidated under MDL 2768 before Judge Indira Talwani for the District of Massachusetts. Lawsuits are also currently being accepted from individuals who suffered complications due to Stryker’s Tritanium Acetabular Shells.
Settlement negotiations continue between the Stryker legal team and plaintiffs pursuing their cases individually as well as in the pending MDLs. You can also file a Tritanium Acetabular Shell lawsuit through a Stryker hip replacement lawyer. The latest Stryker lawsuit update includes:
As of January 2026, two separate multidistrict litigations (MDLs) involving Stryker hip replacements remain open in federal court. Each MDL focuses on different Stryker models, but plaintiffs in both allege design defects that caused similar injuries.
MDL No. 2768 (District of Massachusetts)
MDL No. 2441 (District of Minnesota)
Despite large-scale settlements, both MDLs remain open, and attorneys continue to review and file new cases.
Although most Stryker hip lawsuit claims have been resolved through settlements, attorneys continue to accept new hip replacement lawsuits from individuals who may not have previously joined the litigation.
While both MDLs have been active for many years and now involve fewer pending cases, lawyers continue to investigate and accept new claims, particularly those involving the LFIT V40 femoral head devices.
No major court rulings or procedural changes were reported during this period. However, attorneys continued filing and evaluating individual Stryker hip cases outside the main settlement framework.
MDL 2441 is continuing to shrink through settlement-related dismissals
Attorneys continued pursuing individual cases involving the Tritanium Acetabular Shell.
Several cases were transferred into MDL 2768 from state courts. One newly filed case alleged the need for revision surgery and elevated cobalt and chromium levels, attributed to alleged device defects.
Litigation is ongoing against the Tritanium Acetabular Shell from Stryker, with patients complaining that the manufacturing defects cause several health issues and injuries.
Previously settled cases continued to be formally dismissed from both MDLs as part of ongoing case management.
Attorneys are instructed to provide the court with a lawsuit update about any outstanding cases in the confidential settlement program.
In MDL 2441, Judge Frank set a September 9, 2022, deadline for attorneys to report on unresolved claims remaining in the confidential settlement program.
Multiple cases in MDL 2441 were dismissed with prejudice after plaintiffs were included in settlement agreements.
Order issued stating that discovery is postponed in the cases until January 2022 on account of the confidential settlement program.
On September 9, the MDL court was informed that a settlement agreement had been reached, and the court stayed discovery to allow implementation of the settlement program.
The court set up a formal registration process for unresolved/previously unregistered claims tied to the settlement program, with submissions beginning June 19, 2020.
On November 2, parties in the Stryker Confidential LFIT V40 MDL reported that they reached a private settlement agreement following the FDA-listed recall involving 42,519 units.
On April 5, the Judicial Panel on Multidistrict Litigation (JPML) created MDL No. 2768 (In re: Stryker LFIT Anatomic CoCr V40 Femoral Head Products Liability Litigation) in the District of Massachusetts. It addresses femoral head defects. (62 pending cases as of January 2026).
On November 3, Stryker publicly announced a settlement program for eligible U.S. patients who had revision surgery before November 3, 2014, for reasons related to the voluntary recall.
On June 14, 2013, the JPML consolidated federal lawsuits involving Stryker Rejuvenate and ABG II modular implants into MDL No. 2441 in the United States District Court for the District of Minnesota. This MDL addresses Rejuvenate and ABG II implant defects (36 cases pending as of January 2026).
Your product liability lawsuit against Stryker must be filed within the statute of limitations. This is a concrete time frame for filing lawsuits. Your claim for compensation will be dismissed if it’s not filed within this time.
The statute of limitations differs for every state, but it’s typically up to four years for product liability cases. You’ll need to consult with a lawyer to find out what the time limit is for your particular case.
The MDL judge ruled that those who haven’t had a revision surgery yet to remove the defective Stryker product, and subsequently have one in the future, will be allowed to have the statute of limitations suspended. So even though the initial settlement was in 2014, eligible patients may still be eligible to file a product liability lawsuit and recover compensation from the company.
Given the complicated nature of litigation against Stryker and the various devices involved, it’s best to seek the professional opinion of a lawyer experienced in multi-district litigation to set yourself up for a big settlement.
It’s important to first confirm your eligibility to file a lawsuit. You’re likely eligible to file the lawsuit if you’ve suffered complications after receiving a Stryker implant, and also meet the following –
Basically, you may be able to qualify to file a lawsuit related to your Stryker hip implant if you received one of these implants and are having significant issues.
The best way to create a robust foundation for your lawsuit is to follow a proven legal process to recover compensation from Stryker. This involves the following steps:
At Rosenfeld Injury Lawyers, we have over two decades of experience in conducting multi-district litigation against major corporations like Stryker and have won more than $400 million in settlements for our clients. The mass tort lawyers at our law firm will diligently pursue your case to ensure you get a fair settlement.
It’s difficult to figure out on your own whether the statute of limitations is up for your claim, where the lawsuit must be filed, and how to best prepare the case to get a big Stryker hip settlement.
There’s a much simpler way for you to secure justice and be properly compensated for all of your pain and suffering. The recovery process will go smoothly once you have dedicated product liability lawyers fighting your case.
Hire our lawyers with a demonstrated track record of winning big settlements for clients in complicated multi-district litigation. You’ll have peace of mind that your lawsuit is being handled by professionals who will make sure all legal and medical facts are established under prevailing laws.
Many plaintiffs who agreed to accept the initial Stryker hip settlement offer have already received their payouts. The vast majority of claimants accepted the offer, while settlements for those who didn’t accept the offer were obtained by suing Styker separately from the MDL. New lawsuits filed against the company now are going to take time.
Even if Stryker enters into settlement conferences before individual trials occur, it may take considerable time for plaintiffs to receive a payout, likely a couple of years at the very least. Those who take their case to trial may have to wait for even longer, as factors such as discovery, filings, and negotiations can affect the timeline.
The last thing you need to worry about after being injured by a defective Exactech product is how you’ll pay for a lawyer to get the compensation you deserve. Fighting such a big company isn’t going to be cheap, and you need all the legal assistance you can get. Our mission is to provide access to justice for all those who’ve been affected by these defective implants.
Our law firm is taking Exactech recall cases on a contingency fee basis. This means you don’t have any out-of-pocket costs when hiring a personal injury attorney to file your lawsuit. You’ll continue to benefit from our professional legal representation throughout the entire process without needing to pay us anything.
Our fee is only a percentage of your Stryker hip settlement, so we work hard to ensure you win maximum compensation, and we won’t get paid until you do.
You may be entitled to seek compensation for economic and non-economic losses, as well as wrongful death damages, if someone you love passed away. The facts of your case largely determine the type of damages you can claim.
A lot of Stryker lawsuits have already been settled, but many are still pending while new cases continue to be filed. The settlement amounts will vary depending on the nature of the case and specific facts, as well as other factors, including:
There have been many Stryker hip recall lawsuit settlements over the past ten years. The company first offered a global settlement in 2014 for $1 billion for patients who underwent revision surgery related to the Rejuvenate and ABG II systems, with the total outlay being increased significantly over the next couple of years. Notable Stryker hip lawsuit settlements include:
In multi-district product liability cases, there is no fixed amount that’s paid in damages. The payout is different for each plaintiff, depending on the facts unique to their case. This is true for both pending individual lawsuits as well as any new lawsuits being filed now.
A fixed amount will be provided to those who take a global settlement, like the one Stryker offered in 2014, subject to certain exceptions. Eligible patients who qualified under the Stryker settlement for receiving defective Rejuvenate or ABG II hip systems (and underwent a revision surgery by November 3, 2014) were provided a basic payment of $300,000. In this case, eligible patients had to submit their claim by March 2, 2015. This amount changes if they had to take certain reductions
For example, the damage award will be reduced by 15% if the claimant had another implant before getting both the Rejuvenate devices or the ABG II hip. Stryker has also settled many LFIT V40 Femoral Head lawsuits directly with the plaintiffs for an undisclosed amount.
The more recent settlements have mostly been confidential, so it’s difficult to say what patients should expect. Settlement values for Stryker cases vary, though the early settlements have been less than they have been for other hip replacement cases.
People injured by these implants are filing lawsuits against Stryker for being negligent in the design of its hip devices and releasing the implant devices when they were defective at the time of manufacturing.
Some lawsuits claim the company didn’t act proactively enough once it was known these devices were defective. These premature failures, at higher-than-expected rates, have caused medical issues and injury to patients. Some patients have even been required to or will need to have surgery to replace defective hip implants.
Stryker Corporation is a major medical technology company based in the United States that designs, manufactures, and sells a large variety of orthopedic products. Stryker has provided various hip implant systems for the reconstruction market globally, including the Rejuvenate and ABG II systems. These products are used in surgical procedures like joint replacement. Its products have been implanted in tens of thousands of patients both in the US and other countries.
Most of the problems associated with Stryker Rejuvenate and ABG II modular-neck joint hip stem systems are related to the metal neck components in the implants made of cobalt, chromium, and titanium.
Due to the modular neck design of the Stryker hip implant, the friction caused by the regular movement of these metal-on-metal Stryker hip replacement systems sheds metallic debris into the blood, leading to metal poisoning as well as the following problems:
Metallosis is caused when cobalt and chromium ions are released into the bloodstream and surrounding tissue, causing metal poisoning. Common symptoms of metallosis can include painful inflammation, tissue and bone death, severe pain, and systemic issues, such as problems with the kidney and heart. It can also cause severe symptoms like vision loss, hearing loss, cognitive impairment, and heart failure.
Defective implants become unreliable over time and may cause dislocations, leading to mobility issues for the patient.
Chemical reactions people have from the metal debris going into the surrounding tissue can lead to adverse local tissue reactions, like necrosis, osteolysis (bone degeneration, which can lead to broken bones), and pseudotumors near the site of the implant.
If you’ve received one of these implants and you are dealing with grinding, popping sounds, limited mobility, or discrepancies in leg length, you may be experiencing an implant failure and should reach out for help as soon as possible.
There’s one main defendant in these lawsuits, primarily responsible for the negligent development of these hip replacement devices implanted in thousands of patients across the US.
Stryker designs, manufactures, and sells hip replacement products after obtaining FDA approval. The products are made at its own factory, so there’s no third-party manufacturer to blame for the defects.
There are primarily four product families from Stryker mentioned in lawsuits against the company. If you have received any one of these products as an implant for your hip joint replacement surgery, you’re likely eligible to get compensation.
Stryker Sales Corporation first notified surgeons about a low failure rate of its hip replacement implants due to metallic particles in 2010. The metal-on-metal friction was causing the failures.
As adverse event reports continued to pile up at the FDA, Stryker voluntarily recalled two of its widely used hip replacement products. A recall for the other devices named in these lawsuits has been issued as well.

As your attorneys, we’ll ensure you are fairly compensated for the pain and suffering you’ve endured because of the defective hip implant. Our decades of experience in handling multi-district litigation and solid track record of winning major settlements speak for themselves, and we’ll put this experience to good use for your case.
We’ll take your case on a contingency fee basis, so you get a highly experienced Stryker hip replacement attorney at no upfront cost. You won’t have to pay us anything until you have a settlement you’re happy with.
Secure a free consultation for your Stryker hip recall case by giving us a call today, and our personal injury attorneys will be happy to speak with you to offer detailed advice and any lawsuit information you want to know. You can learn more about all of the legal options available to you and what your settlement offer may look like.
All content undergoes thorough legal review by experienced attorneys, including Jonathan Rosenfeld. With 25 years of experience in personal injury law and over 100 years of combined legal expertise within our team, we ensure that every article is legally accurate, compliant, and reflects current legal standards.